HOME / NEWS / Industry News / Most Reliable Medical Swabs for PCR Testing: Sterile & Polyester Options
Content
- 1 What Makes a Medical Swab Reliable for PCR Testing?
- 2 Swab Tip Materials Compared: Polyester, Nylon Flock, and Rayon
- 3 Polyester Swabs: Properties and PCR Performance
- 4 What Is a Sterile Swab Stick and Why Does Sterility Matter?
- 5 Regulatory and Quality Standards for PCR Swabs
- 6 Selecting the Most Reliable Swab for Your PCR Application
What Makes a Medical Swab Reliable for PCR Testing?
For PCR (polymerase chain reaction) testing to deliver accurate results, the swab used to collect the specimen is just as critical as the laboratory analysis itself. A reliable medical swab for PCR must do three things consistently: collect an adequate volume of biological material, release that material efficiently into transport media, and introduce zero inhibitors that could interfere with the amplification reaction.
PCR is an extraordinarily sensitive technique — capable of detecting a single copy of a target nucleic acid sequence — which means any contamination, nuclease activity, or chemical inhibitor introduced by a low-quality swab tip can produce false-negative or false-positive results. Studies evaluating nasopharyngeal swab performance have shown that tip material, fiber geometry, and adhesive or binder chemistry in the tip construction all measurably affect DNA and RNA elution efficiency, with differences of 30–50% in elution yield documented between poorly performing and optimized swab designs.
The most commonly evaluated swab types for PCR specimen collection include flocked nylon, polyester, and rayon-tipped swabs. Each has distinct characteristics that make it more or less suitable depending on the specimen site, the PCR assay platform, and the transport system in use.
Swab Tip Materials Compared: Polyester, Nylon Flock, and Rayon
Not all swab tips perform equally in PCR workflows. The fiber type, tip architecture, and binder system determine how cells and viral particles are captured at the collection site and how completely they are released into the extraction tube.
| Tip Material | Collection Efficiency | Elution into Transport Media | PCR Inhibitor Risk | Typical Applications |
|---|---|---|---|---|
| Flocked Nylon | Highest — perpendicular fibers act as micro-bristles | Excellent — up to 95% release | Very low | Nasopharyngeal, SARS-CoV-2, influenza, STI PCR |
| Polyester | Good — woven fiber matrix captures material well | Good — 70–85% typical elution | Very low — no natural binders or fatty acids | Throat, wound, urethral, environmental swabs; viral transport |
| Rayon | Moderate | Moderate — fibers retain more material | Low-moderate — some lots contain PCR inhibitors | Culture-based microbiology; less preferred for PCR |
| Cotton | Moderate | Poor — fibers trap nucleic acids | High — fatty acids inhibit Taq polymerase | Not recommended for PCR |
Cotton swabs — despite their widespread general use — are contraindicated for PCR-based diagnostics. Natural cotton fibers contain fatty acids and other organic compounds that inhibit Taq DNA polymerase, the enzyme that drives PCR amplification. Multiple validation studies, including guidance published by the CDC and WHO during the COVID-19 pandemic, explicitly excluded cotton-tipped swabs from approved specimen collection devices for SARS-CoV-2 molecular testing.
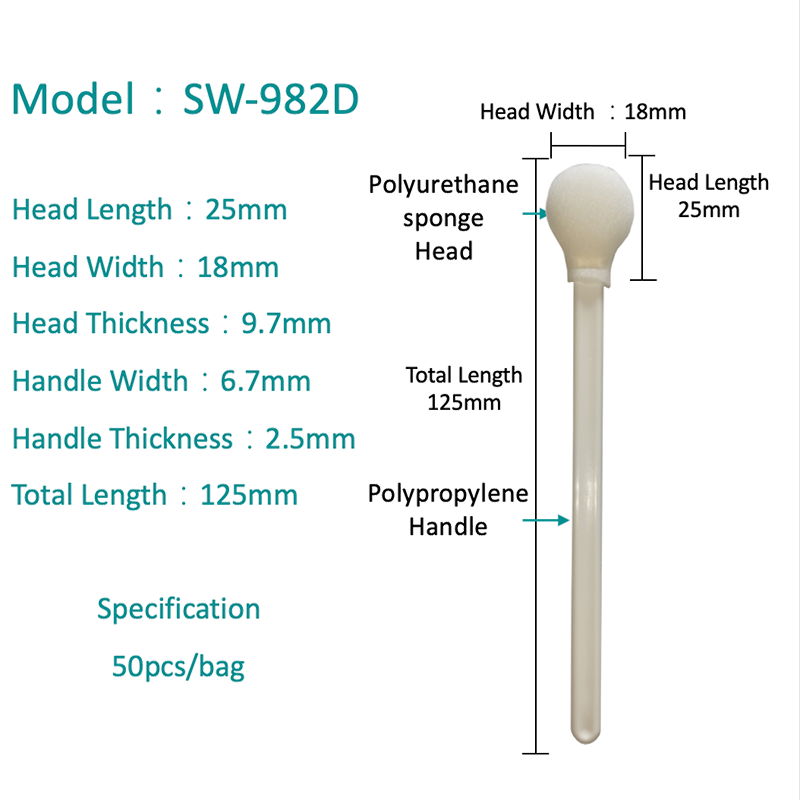
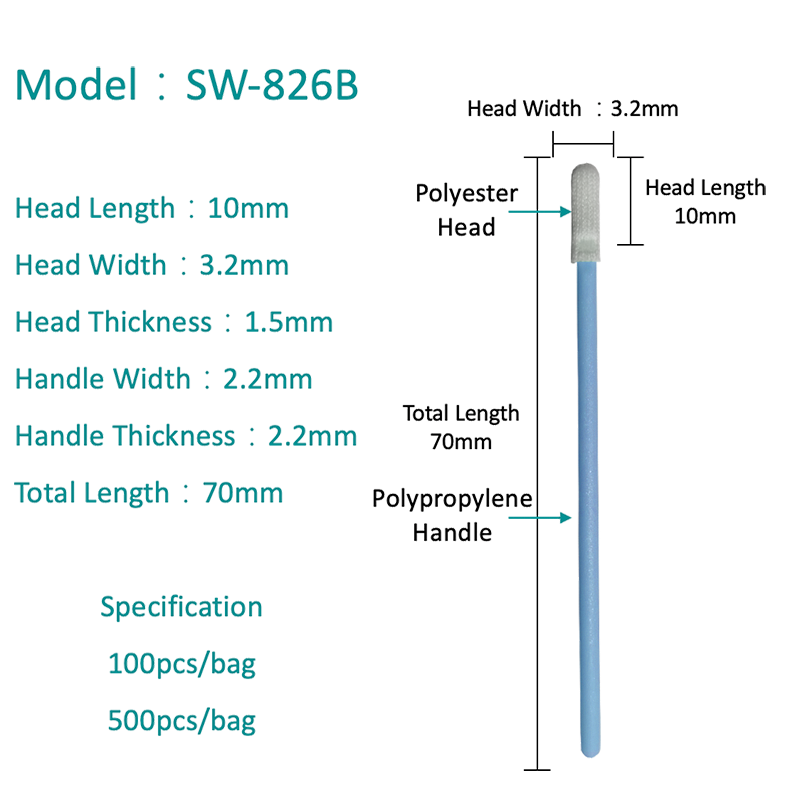
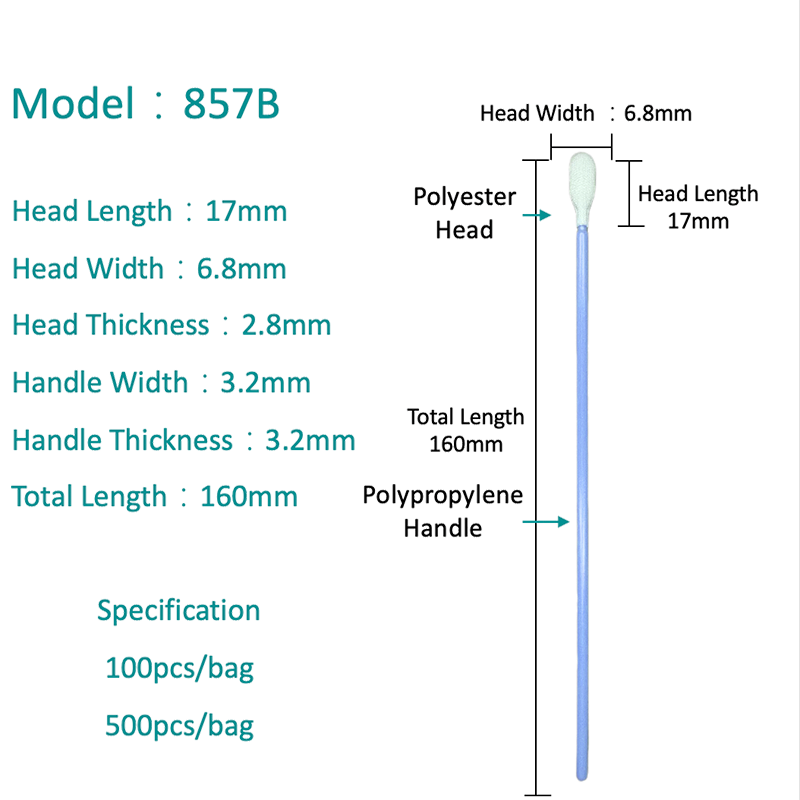
Polyester Swabs: Properties and PCR Performance
A polyester swab consists of a woven or knit polyester fiber tip bonded to a plastic or wire shaft. Polyester is a fully synthetic material with no natural organic compounds, making it inherently free of PCR inhibitors — one of its most important advantages in molecular diagnostics.
Key Advantages of Polyester Swabs
- Chemical inertness: Polyester does not leach substances that interfere with nucleic acid extraction or amplification. This makes polyester swabs compatible with all major viral transport media (VTM), universal transport media (UTM), and dry swab PCR protocols.
- Low background DNA: High-quality polyester swabs manufactured under cleanroom conditions carry negligible background human or microbial DNA, reducing the risk of contamination artifacts in sensitive assays.
- Good absorptive capacity: The woven fiber structure absorbs and holds liquid samples effectively at the collection site. Unlike flocked swabs, polyester tips retain more of the specimen within the fiber matrix — which is advantageous in transport but requires thorough vortexing during elution to achieve complete release.
- Chemical and heat stability: Polyester fibers are stable across the pH and temperature ranges encountered in specimen transport and storage (−80°C frozen archive to ambient transport conditions), maintaining sample integrity from collection to laboratory analysis.
- Broad specimen site compatibility: Polyester swabs are validated for throat, nasal, nasopharyngeal, wound, urogenital, rectal, and environmental surface sampling — making a single swab format adaptable across multiple test menus in a clinical laboratory.
Optimizing Elution from Polyester Swabs
Because polyester fiber tips retain more material than flocked tips, laboratory protocols using polyester swabs should include vigorous vortexing for 15–30 seconds after immersion in transport media, followed by manual expression of the swab tip against the tube wall before discarding. This step significantly improves cell and viral particle release and reduces variation between specimens. Some automated extraction platforms include dedicated swab elution cycles optimized for polyester tip geometry.
What Is a Sterile Swab Stick and Why Does Sterility Matter?
A sterile swab stick is a specimen collection device — tip plus shaft — that has been processed to eliminate all viable microorganisms, spores, and nucleic acid contamination prior to use. Sterility is not a general term in medical swab manufacturing; it is a defined, validated state achieved through gamma irradiation, ethylene oxide (EtO) gas sterilization, or electron beam irradiation, and verified through sterility testing per pharmacopoeial standards (USP <71>, EP 2.6.1).
Sterility Requirements by Application
- Clinical PCR diagnostics: Swabs must be sterile and DNase/RNase-free. Residual nucleases on a non-sterile swab can degrade RNA targets — critical for RNA virus detection (influenza, SARS-CoV-2, RSV) — producing false-negative results even from well-collected specimens.
- Wound and surgical site sampling: Requires microbiologically sterile swabs to avoid introducing exogenous organisms that could confound culture results or — in the case of open wounds — cause secondary infection.
- Sexually transmitted infection (STI) testing: Urogenital and rectal swabs used in STI molecular panels must be sterile and free of human DNA background to avoid cross-contamination in high-sensitivity PCR assays targeting Chlamydia, gonorrhoea, and Mycoplasma.
- Environmental monitoring: Swabs used to sample pharmaceutical manufacturing surfaces for microbial contamination require a defined sterility assurance level (SAL) of 10⁻⁶ — one chance in one million of a viable organism being present on any single swab.
Shaft Materials for Sterile Swab Sticks
The shaft of a sterile swab stick must be compatible with the sterilization method and the collection site anatomy. The main options are:
- Polystyrene or polypropylene plastic shafts: The most common shaft material for PCR swabs. Rigid, lightweight, available in standard lengths (15–20 cm) and breakpoint formats where the shaft snaps at a defined point to fit into a standard transport tube. Fully compatible with gamma and EtO sterilization.
- Flexible wire (aluminum core): Used for nasopharyngeal swabs that must navigate the nasal anatomy to the posterior nasopharynx. The wire shaft is malleable, allowing the clinician to pre-bend the swab for patient anatomy, and is coated with nylon or plastic to prevent tissue abrasion.
- Wood (birch wood sticks): Used in some culture-based bacteriology swabs but not suitable for PCR — wood contains tannins and other polyphenols that are potent PCR inhibitors and are not removed by sterilization.
Regulatory and Quality Standards for PCR Swabs
Medical swabs used in diagnostic PCR workflows are regulated as in vitro diagnostic (IVD) medical devices in most jurisdictions. Buyers — clinical laboratories, hospital procurement teams, diagnostic kit manufacturers — should verify that their swab supplier meets applicable regulatory and quality requirements:
- ISO 13485:2016 — Quality management system standard for medical device manufacturers. Ensures design controls, production controls, and post-market surveillance processes are in place.
- EU MDR 2017/745 / IVDR 2017/746 — European regulatory framework for medical devices and IVD devices. CE marking under IVDR is required for swabs marketed as IVD specimen collection devices in the EU.
- FDA 510(k) clearance or EUA authorization — In the United States, swabs intended for use in FDA-cleared IVD test systems may require 510(k) clearance or demonstration of compatibility with cleared collection systems. During COVID-19, emergency use authorizations specified acceptable swab types for authorized SARS-CoV-2 tests.
- DNase/RNase-free certification: Suppliers should provide lot-level certificates of analysis confirming absence of nuclease activity — typically tested by incubating a swab eluate with a synthetic RNA or DNA substrate and confirming no degradation after a defined time at 37°C.
- Human DNA-free certification: Particularly important for swabs used in forensic applications and highly multiplexed PCR panels. Verified by running swab eluate through a validated human DNA quantification assay (e.g., Quantifiler Trio) and confirming results below the assay's limit of detection.
Selecting the Most Reliable Swab for Your PCR Application
The right swab choice depends on the specimen site, the PCR assay system, and the transport and storage conditions. Here is a practical decision framework:
- Check your assay's Instructions for Use (IFU). PCR kit manufacturers validate their assays with specific swab types and transport media. Using an unlisted swab type may invalidate assay performance claims and regulatory compliance. Always start with the swab types listed in the IFU as acceptable.
- Prioritize flocked nylon for nasopharyngeal PCR. Flocked swabs consistently outperform wound-fiber swabs for nasopharyngeal and anterior nasal specimen collection in respiratory virus PCR, due to superior cell collection and elution efficiency.
- Use polyester swabs for throat, wound, and urogenital sites. Polyester swabs offer an excellent balance of collection capacity, elution performance, and chemical inertness — and are typically more cost-effective than flocked swabs for high-volume routine testing.
- Always specify sterile, DNase/RNase-free for RNA virus targets. For influenza, RSV, SARS-CoV-2, and other RNA targets, nuclease-free certification is non-negotiable. Request lot-specific certificates of analysis from your supplier.
- Validate before switching suppliers. If changing swab brand or batch during an established PCR workflow, perform a bridging validation comparing sensitivity and specificity of results between the existing and new swab — particularly for assays operating close to their limit of detection.
Specimen collection is the first and most variable step in any PCR testing workflow. Investing in validated, high-quality sterile swabs — whether polyester, flocked nylon, or a site-specific format — directly protects the clinical value of every PCR result that follows.




 English
English  中文简体
中文简体