HOME / NEWS / Industry News / Cheek Swab DNA Testing, Mouth Swab STD Tests & Buccal Swab Guide
A buccal or cheek swab is the preferred method for DNA collection and oral STD testing because it is completely non-invasive, painless, and highly accurate. The inner cheek contains stratified squamous epithelial cells that are rich in nuclear DNA, making them ideal for genetic analysis, paternity testing, and forensic identification. For STD detection, throat swabs using nucleic acid amplification tests can reliably identify chlamydia and gonorrhea infections acquired through oral sex. The procedure never causes bleeding because the epithelial tissue of the buccal mucosa is avascular, meaning it contains no blood vessels within the tissue layer being sampled. Microfiber polyester swabs have emerged as superior collection tools compared to traditional cotton, offering higher DNA recovery rates, better sample release characteristics, and reduced PCR inhibitor contamination.
Content
- 1 Why a Buccal Swab Is Used to Collect DNA
- 2 Mouth Swab STD Tests: Detecting Oral Infections
- 3 Why a Buccal Swab Procedure Should Not Cause Bleeding
- 4 Microfiber Polyester Swabs: Advantages Over Traditional Materials
- 5 Best Practices for Buccal Swab Collection and Handling
- 6 Applications and Future Directions in Swab-Based Diagnostics
Why a Buccal Swab Is Used to Collect DNA
Buccal swabs have become the standard for DNA collection across clinical diagnostics, genetic research, forensic science, and direct-to-consumer ancestry testing. The inner cheek provides an abundant source of nucleated epithelial cells that contain high-quality genomic DNA suitable for PCR amplification, microarray genotyping, and next-generation sequencing.
Non-Invasive and Patient-Friendly Collection
Unlike blood draws that require trained phlebotomists, needles, and clinical infrastructure, buccal swab collection can be performed by the subject themselves with minimal instruction. This self-collection potential dramatically reduces costs, eliminates needle phobia barriers, and enables large-scale population studies including children and elderly participants who may resist venipuncture. Studies have validated that cytobrush buccal swabs provide sufficient DNA yield for most genotyping applications with collection times as short as 15 seconds.
DNA Yield and Quality for Genetic Analysis
While blood samples yield higher absolute quantities of DNA, buccal cells provide ample material for modern genotyping methods that require only 2 to 10 nanograms per assay. The DNA extracted from buccal swabs supports PCR-based testing, STR profiling for forensics, SNP analysis for disease risk assessment, and genome-wide association studies. DNA stability at room temperature simplifies transport and storage, making buccal swabs practical for multicentric studies and mail-based collection protocols.
Cost Efficiency and Scalability
Buccal swab collection is significantly more cost-effective than blood collection when accounting for personnel, equipment, and processing expenses. The final cost per sample for cytobrush collection and extraction averages approximately 4.66 euros compared to 7.17 euros for mouthwash protocols, while providing comparable DNA quality. This economic advantage becomes critical when studies require thousands of samples, making buccal swabs the practical choice for biobanking and large epidemiological research.
Mouth Swab STD Tests: Detecting Oral Infections
Mouth swab STD tests are essential for detecting sexually transmitted infections that localize in the throat or oral cavity, particularly chlamydia and gonorrhea transmitted through oral sex. Standard urine tests cannot detect these oral infections, making targeted swab collection the only reliable diagnostic method.
Infections Detected by Oral Swab Testing
Throat swab testing primarily targets Chlamydia trachomatis and Neisseria gonorrhoeae, the bacteria responsible for chlamydia and gonorrhea respectively. These infections often present asymptomatically in the throat, meaning carriers can unknowingly transmit them to partners through kissing or oral sex. Comprehensive oral STD panels may also test for syphilis, herpes simplex virus types 1 and 2, trichomoniasis, Mycoplasma genitalium, and Ureaplasma urealyticum depending on the laboratory offering.
Nucleic Acid Amplification Test Technology
Modern mouth swab STD tests use nucleic acid amplification technology, or NAAT, which detects the genetic material of pathogens with exceptional sensitivity and specificity. NAAT can identify infections even when bacterial loads are low, achieving accuracy rates comparable to or exceeding culture-based methods. The CDC reports over 2.5 million combined cases of chlamydia, gonorrhea, and syphilis annually in the United States, with chlamydia accounting for 1.6 million diagnoses, underscoring the critical need for accurate testing.
Collection Technique and Timing
Proper throat swab collection requires rubbing the posterior pharynx and tonsillar arches without touching the cheeks, tongue, or teeth to avoid contamination. Patients should avoid eating, drinking, smoking, chewing gum, or brushing teeth for at least 30 minutes prior to collection. The swab is then placed in transport medium to preserve pathogen viability during transit to the laboratory. Results typically return within 2 to 3 business days for laboratory-processed samples.
At-Home Testing Options
FDA-approved at-home collection kits now allow individuals to collect vaginal swabs, throat swabs, or oral fluid samples privately and mail them to certified laboratories. The Visby Medical Women's Sexual Health Test, approved in March 2025, offers PCR-based results for chlamydia, gonorrhea, and trichomoniasis in 30 minutes using a vaginal sample. For oral infections, self-collection throat swab kits provide discreet testing alternatives that reduce barriers to screening.
| Method | Invasiveness | DNA Yield | Best Use Case |
|---|---|---|---|
| Buccal Swab | Non-invasive | Moderate to High | Genetic testing, forensics, ancestry |
| Blood Draw | Invasive | Very High | High-volume genotyping, diagnostics |
| Mouthwash | Non-invasive | High | Research studies, microbiome analysis |
| Throat Swab STD | Minimally invasive | N/A | Oral chlamydia and gonorrhea detection |
Why a Buccal Swab Procedure Should Not Cause Bleeding
The buccal swab procedure is intentionally designed to be painless and bloodless, a feature rooted in the fundamental anatomy and histology of the oral mucosa. Understanding why bleeding does not occur requires examining the tissue structure of the inner cheek and the mechanics of cell collection.
Avascular Nature of Epithelial Tissue
The buccal mucosa is lined with stratified squamous epithelium, a tissue type that is avascular, meaning it contains no blood vessels within the epithelial layer itself. Blood vessels supplying the oral mucosa are located in the underlying connective tissue layer called the lamina propria, separated from the surface by the basement membrane. When a swab gently rubs the inner cheek, it only dislodges superficial epithelial cells from the apical layers of the epithelium. Because the swab never penetrates through the basement membrane into the vascularized connective tissue beneath, no blood vessels are damaged and bleeding cannot occur.
Tight Junctions and Superficial Cell Removal
Epithelial cells are bound together by tight junctions and desmosomes that create a cohesive barrier. The gentle friction applied by a buccal swab is sufficient to slough off only the outermost dead or dying cells that are already destined for natural shedding. This superficial removal does not disrupt the deeper living cell layers or the basement membrane anchoring the epithelium to underlying tissues. The force required to cause bleeding would need to lacerate through multiple cell layers and the basement membrane, which is far beyond the pressure exerted during standard swab collection.
Protective Features of the Oral Mucosa
The oral cavity is evolutionarily adapted to withstand mechanical abrasion from chewing and speaking. The buccal mucosa is relatively thick, resilient, and continuously regenerates its surface layers. Saliva provides additional lubrication that reduces friction during swab contact. These protective mechanisms ensure that the mild abrasion from a soft swab tip causes no trauma, inflammation, or vascular injury, making the procedure safe for all age groups including infants and elderly patients with fragile tissues.
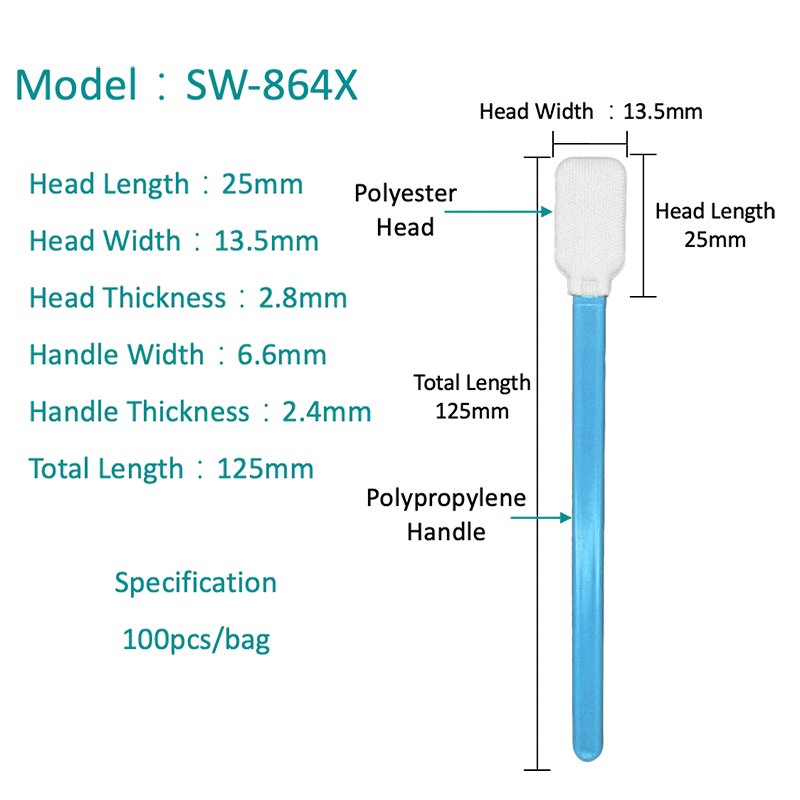
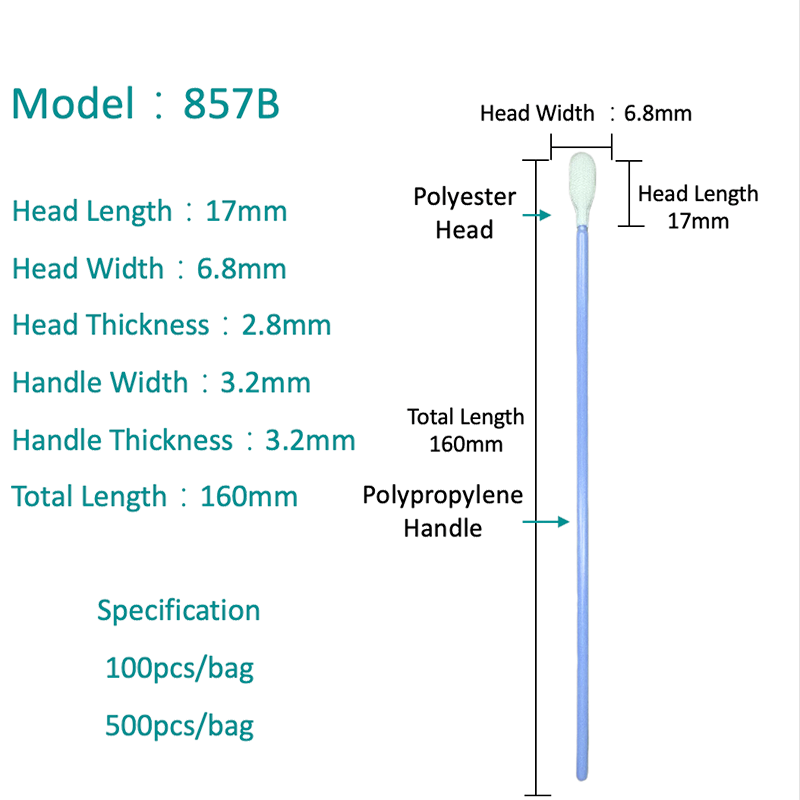
Microfiber Polyester Swabs: Advantages Over Traditional Materials
Microfiber polyester swabs represent a significant advancement in biological sample collection technology, offering measurable improvements in DNA recovery, sample release efficiency, and analytical compatibility compared to traditional cotton swabs. These synthetic swabs are engineered specifically for molecular diagnostics and forensic applications where sample integrity is paramount.
Enhanced DNA Collection and Release
Microfiber polyester swabs feature ultra-fine synthetic fibers that create a high surface area for capturing epithelial cells and biological fluids. Unlike cotton, which can trap samples within its fibrous matrix, polyester microfibers release collected material more efficiently during extraction. This improved elution increases DNA yields and reduces the loss of genetic material during laboratory processing. Studies comparing swab types for forensic DNA collection have shown that synthetic fiber designs can outperform cotton in specific recovery scenarios, particularly when sample quantities are limited.
Reduced PCR Inhibitor Contamination
A critical disadvantage of cotton swabs is the presence of fatty acids and other natural compounds that can inhibit PCR amplification, potentially causing false-negative results or degraded DNA profiles. Microfiber polyester swabs are manufactured from pure synthetic polymers that do not introduce these biological inhibitors. This chemical inertness ensures that collected samples remain compatible with sensitive molecular assays including quantitative PCR, STR profiling, and next-generation sequencing without requiring additional purification steps to remove contaminants.
Durability and Sterility Standards
Polyester microfibers offer superior durability and resistance to chemicals compared to natural fibers. Swab batches are routinely treated with ethylene oxide sterilization and tested for human DNA contamination to ensure forensic-grade purity. The synthetic construction resists shedding during collection, preventing fiber fragments from interfering with extraction protocols or contaminating laboratory equipment. These characteristics make microfiber polyester swabs the preferred choice for high-stakes applications including forensic evidence collection, paternity testing, and clinical diagnostics where sample integrity directly impacts case outcomes.
Comparative Performance in Forensic Studies
Research evaluating swab materials for forensic DNA testing has demonstrated that performance varies by substrate and sample type. On glass surfaces, cotton swabs have shown advantages for blood collection, while polyester swabs excel at collecting touch DNA from textured plastic surfaces commonly found in vehicles. For buccal cell collection, flocked swabs with bicomponent fibers and microfiber designs consistently demonstrate higher DNA recovery and better STR profiling success rates compared to standard cotton applicators. The choice between swab types should align with the specific application, but microfiber polyester provides the most versatile performance across diverse collection scenarios.
Best Practices for Buccal Swab Collection and Handling
Achieving reliable results from cheek swab DNA testing or mouth swab STD tests depends on proper collection technique, appropriate swab selection, and correct handling procedures. Deviations from established protocols can lead to sample degradation, contamination, or insufficient material for analysis.
Pre-Collection Preparation
Subjects should refrain from eating, drinking, chewing gum, smoking, or brushing teeth for at least 30 minutes before sample collection. These activities can introduce food particles, bacteria, and chemical residues that contaminate samples or inhibit PCR reactions. For DNA collection, rinsing the mouth with water immediately before swabbing can reduce bacterial load without affecting human cell yield.
Collection Technique for Maximum Yield
For buccal DNA collection, firmly rub the swab against the inner cheek for 15 to 60 seconds, rotating the swab to expose all surfaces to the mucosa. For STD throat swabs, wipe the posterior pharynx and tonsillar arches back and forth while avoiding contact with the tongue, teeth, and cheeks. The swab tip should not touch any surfaces other than the target collection site to prevent environmental contamination.
Storage and Transport Requirements
Immediately after collection, place the swab into the provided collection tube or transport medium. DNA swabs can remain stable at room temperature for extended periods, though refrigeration or freezing at minus 20 degrees Celsius is recommended for long-term storage. STD swabs must be placed in appropriate transport media to preserve pathogen viability and should reach the laboratory within the timeframe specified by the test manufacturer, typically within 24 to 72 hours.
Applications and Future Directions in Swab-Based Diagnostics
Buccal swab technology continues to expand beyond traditional DNA banking into emerging diagnostic fields. Epigenetic age prediction using DNA methylation patterns from buccal cells has achieved surprising accuracy, with researchers developing models that account for the cellular composition of buccal samples to improve precision. In conservation genetics, buccal swabs enable non-destructive DNA collection from endangered species, with studies demonstrating successful long-term storage at room temperature for over 18 months while maintaining amplification capability.
The integration of microfiber polyester swabs with direct PCR workflows is streamlining forensic analysis by eliminating extraction steps and reducing processing time. As synthetic swab materials advance and collection protocols become more standardized, the accessibility and accuracy of swab-based diagnostics will continue to improve, supporting broader population screening, personalized medicine, and rapid-response public health surveillance.




 English
English  中文简体
中文简体